WHAT ARE EUROTHREADS?
NO CUTS OR INCISIONS. PURELY INJECTIONS!
EuroThreads are resorbable surgical sutures composed of biodegradable polymers. The treatment is minimally invasive and produces instant results. Polydioxanone (PDO) sutures are used for biomedical applications, particularly in cardiothoracic surgery because it is fully absorbed by hydrolysis within 4 to 6 months and doesn't create any scar tissue. The foreign body stimulates collagen synthesis and the skin structure created will hold for 12 to 15 months.


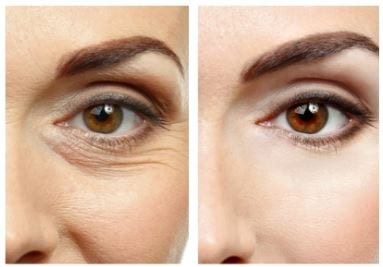
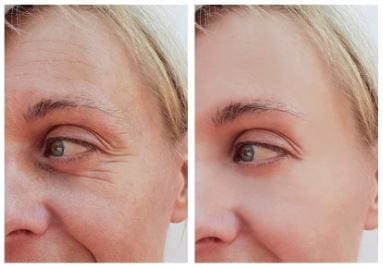

How long has thread lifting been available?
Originally Contour threads received FDA approval in early 2006 which was quickly revoked due to the nature of the threads permanence. These threads were not "absorbable" and could not be removed once they were inserted. EuroThreads uses a biodegradable suture that has been FDA approved for decades in cardiovascular surgery. In 2015, the FDA approved the same suture for aesthetic treatments. Thread lifting can be used for treatments to the face, neck and body.
Are PDO threads FDA approved?
Yes, the use of PDO and PLLA threads have been FDA approved as a medical device for use in soft tissue approximation where the implantation of surgical sutures is appropriate. EuroThreads FDA registration can be confirmed by running a search directly on the government issued FDA website under Medical Device Listings. Further, EuroThreads is registered with the CDRH, Center for Devices and Radiological Health as required by the FDA. EuroThreads Establishment registration #10054861. And EuroThreads device registration #3013648997. Make sure that the product that you choose to use has both an "Establishment" and "Device" registration number as shown above.
What are EuroThreads?
EuroThreads are individual biodegradable, surgical sutures that are pre-inserted into a needle or cannula.
How do EuroThreads work?
A single thread is inserted into either a needle or a cannula during the manufacturing process and then sterilized. A single needle or cannula (thread) is individually inserted into the dermis and upon removal, the thread is left in place. Depending on thread type (Smooth, Twisted or Barbed) the thread is either left in place to promote collagen production or is pulled in an intended direction to produce an appearance commonly associated with lifting.
What are the possible complications associated with PDO thread insertion?
Slight chance of infection or bruising at injection site. Possible but rare chance for thread extrusion.
Do not use EuroThreads on oncology patients or patients with Keloid tendency, bleeding disorders, acne.
Can EuroThreads be used with neurotoxins like BOTOX and dermal fillers?
Yes, there are no contraindications that prevent the use of neurotoxins and fillers with EuroThreads. Both neurotoxins and fillers take some time to produce the desired result so this could change or negatively augment any threading results. It is recommended to let the neurotoxins and filler set in before inserting EuroThreads. This will ensure that the patient has the best opportunity to get the maximum effects from both treatments.

Schedule Your Consultation
Find out if EUROTHREADLIFT is for you!
